Unlike essential oils, essential amino acids are a well named category of compounds. Referring to those that we are unable to make for ourselves and must be consumed in the diet, it was this property that was key to the discovery of the last of the twenty common amino acids: threonine.
The Last Essential Amino Acid
By the start of the 20th century it was already known that some proteins were better at nourishing the animal body than others. This was clearly demonstrated in research reported by Osborne and Mendel (not the one who bred pea plants) between 1912 and 1914. They experimented on rats by feeding them on diets whereby their only nitrogen source came from a specific protein supplement. When fed with a protein that was deficient in lysine, the rats would stop growing, and would only continue if lysine was added to their diet. When fed on a protein which was deficient in both tryptophan and lysine, they would simply die, unless tryptophan was returned, and then only grow if lysine was also returned. From this they concluded that the rats were unable to synthesise the compounds for themselves and must get them from their food: they were essential.1–3
And so the experiments continued, requiring proteins to be identified that were lacking in one amino acid or another so that they could be fed to rats to determine if they were essential.4 This was until work began in 1930, led by the American biochemist William C. Rose, to synthesise large batches of all the known amino acids in order to perform some more focused feeding experiments. This time, the rats were fed on a mixture of amino acids modelled on the proportions found in the protein casein, which had known nutritional quality. However, the rats failed to grow. They concluded that there must be an essential amino acid that was, as yet, unknown to science.
A series of purification experiments ensued on large quantities of protein, trying to isolated this unknown compound which could be added to standard amino acid mix and allow the rats to grow as normal, and in 1935 a preparation of thin, hexagonal crystals was identified as that compound.5 All that remained was the determination of the structure.
It was quickly established that the unknown substance was one of the four possible stereoisomers of α-amino-β-hydroxybutyric acid, but it took another year to determine what the exact stereochemistry was.6 The details of how they did this are a bit involved, and not essential to this story, but I think it’s an interesting example of the incredible deduction skills chemists could implement before they had the technology available to us today so I’ve described it at the end of this post (feel free to not scroll down that far). The short version is that after the first stereocentre had been determined to be the same as every other amino acid, there only remained two possibilities and the configuration was either analogous to the sugar threose, or its stereoisomer erythrose. Once it had been established that the former was true, the amino acid was named threonine in homage to its structurally similar cousin.

Threose and Erythrose
While the story to this point describes the last step of the etymological journey, it doesn’t do much to explain where the prefix ‘threo’ actually comes from. For that, we need to explore threose further. This sugar was discovered in 1901 by the German chemist Otto Ruff. He was working on the synthesis of tetrose sugars (those with four carbons) by degradation of pentose sugars (five carbons). Four tetroses were thought to be possible, two of which had been identified: L- and D-erythrose. These could be synthesised from the pentose sugar, arabinose. Ruff’s work focused on the isolation of one of the other tetroses from another pentose sugar, xylose; a feat which he achieved. Due to the close relationship of this new compound to the previously discovered erythrose, he formed a new name simply by rearranging the letters, hence erythrose became threose.7

The final step in this journey is then to explain where the name erythrose came from. This sugar was discovered fifty years before its near-anagram, in 1850. Isolated from rhubarb, the compound was found to have the interesting property of turning red in the presence of alkalis. Therefore is was named erythrose by its discoverer, the French pharmacist Garot, from the Greek word erythraino, meaning to redden.8
Blind Alleys
Determining the etymology of erythrose appeared for a while to be much more complicated than is described above. It turns out that the “erythr” prefix is quite common, and one could be forgiven for assuming (i.e. I forgive myself for assuming) that one was named first and the rest were variations on the theme. This seems particularly fair when three of such compounds are structurally related: erythrose, erythritol and erythrin.
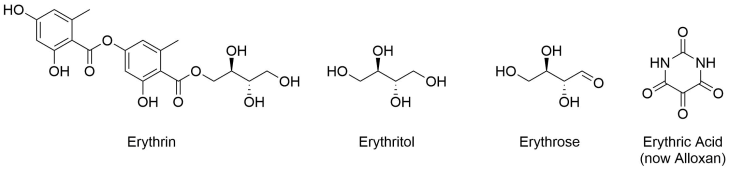
These three compounds share the same core structure, with erythritol being a fully reduced form of erythrose, and erythrin being an ester of erythritol. So, when one finds the paper naming erythrin,9 published 20 years before erythrose,8 and given the simple chemical steps that would take you from erythrin to erythrose, the story seems clear. Except, the authors writing about the discovery of erythrose don’t mention it. For a while I assumed this was an oversight on their part; they must have known that the compounds were related, were inspired by the name and amended the suffix to reflect the sugar-like properties of their new compound. But I simply couldn’t find any evidence of this. Erythrose was isolated from rhubarb. Erythrin was isolated from lichen. The latter was named from a different form of the same Greek work, eryhthros, meaning red, due to the red pigments that could be produced from it (despite being colourless itself and coming from a colourless species of lichen).9,10 Erythritol may have been isolated from erythrin, but it’s difficult to track this one down as it appears to have been known by many other names at the time. Thrown into this mix was an entirely unrelated compound called erythric acid, which was named in 1818 and reassigned its modern name of alloxan in 1838.11,12 It’s no wonder there are multiple variations on the etymology of these compounds in different books and on different websites.
Conclusion
To summarise quite a rambling story: threonine was the last of what we consider to be the common amino acids to be discovered, and were it not for the fact that it is biologically essential, it may have been an even more recent discovery. Unique among the amino acids for possessing two stereocentres, it was this property that was central to generating its name, by analogy to the configuration of the neighbouring hydroxyl groups of threose. Threose, in turn, was named for being an isomer of erythrose by jumbling up the letters (and taking a couple out). It is then erythrose which had what I have come to think of as a traditional etymology, being derived directly from a Greek word, in this case the word for to redden, due to its tendency to blush when basic.
Determining the Stereochemistry of Threonine: The Details
If you’re just in this for the etymology, now is probably the time to stop reading. As I alluded to above, I am continuously fascinated by the lengths chemists of old had to go to to determine the fundamental properties of compounds. Science before NMR must have taken a lot more patience that I could have given it!
The process to determine the configuration of what would be named threonine was published by Meyer and Rose in 1936.6 First, they reduced the alcohol out of a batch of their α-amino-β-hydroxybutyric acid and, despite half racemising the remaining stereocentre, they compared the specific rotation of their product against previously published values for α-amino-β-butyric acid and established the α-carbon possessed the same L-configuration as all the previously isolated amino acids.

This left two possibilities, with relative configurations that were analogous to those of the sugars L-erythrose or D-threose.

By means of an oxidation reaction, followed by treatment with bromine, the new amino acid was converted to lactic acid and isolated as a zinc salt, which gave a positive specific rotation. As it was known that the aqueous zinc salt of natural lactic acid has a negative specific rotation, it was established that the second stereocentre of the new amino acid was of the opposite configuration (D).
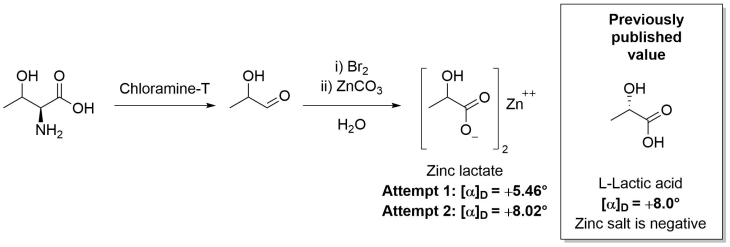
Therefore, the new amino acid was established as having the same configuration as D-threose, and therefore named threonine.

- Osborne TB, Mendel LB. Nutritive properties of maize kernel. J Biol Chem. 1914;18(1):1-16. http://www.jbc.org/content/18/1/1.full.pdf.
- Osborne TB. The Role of Gliadin in Nutrition. J Biol Chem. 1912;12(3):473-510. http://www.jbc.org/content/12/3/473.short.
- Osborne TB, Mendel LB. Amino-acid in nutrition and growth. J Biol Chem. 1914;17(3):325-350.
- Bradford Vickery H. The History of the Discovery of the Amino Acids II. A Review of Amino Acids Described Since 1931 as Components of Native Proteins. Adv Protein Chem. 1972;26:81-171. doi:10.1016/S0065-3233(08)60140-0
- McCoy RH, Meyer CE, Rose WC. Feeding Experiments with Mixtures of Highly Purified Amino Acids. J Biol Chem. 1935;112:283-302.
- Meyer CE, Rose WC. The Spatial Configuration of α-amino-β-hydroxy-n-butyric acid. J Biol Chem. 1936;115(3):721-729.
- Ruff O. Ueber die Oxydation der l‐Arabonsäure und l‐Xylonsäure. Berichte der Dtsch Chem Gesellschaft. 1901;34(2):1362-1372. https://onlinelibrary.wiley.com/doi/10.1002/cber.19010340204.
- Garot. De la matiere colorante rouge des rhubarbes exotiques et indigenes et de son application (comme matiere colorante) aux arts a la pharmacie. J Pharm Chim. 1850;3(17):5-19. https://books.google.co.uk/books?id=2bQ8AAAAcAAJ&pg=PA5&redir_esc=y#v=onepage&q&f=false.
- Heeren F. Untersuchungen Über die Bildung des Flechtenrothes. J fur Chemie und Phys. 1830;59:313-355. https://hdl.handle.net/2027/nyp.33433069069395?urlappend=%3Bseq=333.
- Kane R. Contributions to the Chemical History of Archil and of Litmus. Philos Trans R Soc London. 1840;130:273-324. https://royalsocietypublishing.org/doi/10.1098/rstl.1840.0016.
- Brugnatelli G. Observations of the various changes which take place on treating uric with nitrous acid, and on a new acid called “Erythric” thence produced. Philos Mag J. 1818;52:30-46. https://books.google.co.uk/books?id=knYThj_i9yEC&pg=PA38&lpg=PA38&dq=erythric+acid&source=bl&ots=N6vw8fxiFm&sig=ACfU3U214e3CjGKe5ScQd1jT2gMRTu6mFg&hl=en&sa=X&ved=2ahUKEwiHnaj_xpnlAhUhoVwKHVe9CI0Q6AEwBHoECAgQAQ#v=onepage&q=erythric acid&f=false.
- Wohler F, Liebig J. Untersuchungen über die Natur der Harnsäure. Ann der Chemie und Pharm. 1838;26(241-340):252. https://hdl.handle.net/2027/uva.x002457907?urlappend=%3Bseq=626.

This was a lovelly blog post
LikeLike
This appear to be the most informative content on the web related to the etymology of these names in chemistry.
LikeLike